
by Grow Up Conference | Aug 21, 2024 | Grow Opportunity, Media Partners
By Liam Casey and Allison Jones
Ontario is revamping its approach to the drug crisis by banning consumption sites that are close to schools, introducing a number of treatment hubs and ending the practice of safer supply, Health Minister Sylvia Jones said Tuesday.
The province will shut down 10 consumption and treatment sites – more than half of the provincially funded locations – and critics warned the fundamental shift will lead to more drug poisoning deaths.
Jones denied that the changes would lead to harm.
“People are not going to die. They are going to get access to treatment,” Jones said.
“I do not call watching someone inject an illicit drug to be health care in the province of Ontario. We need to do better, and we can do better.”
The province will introduce legislation in the fall that would prohibit municipalities or organizations from launching new consumption sites or participating in the federal government’s safer supply program that sees prescription medication given to people instead of drugs bought off the street.
Jones said community safety is behind her decisions.
“We need to do more to protect public safety, especially for young school children, while helping people get the treatment they need, which is why we’re taking the next step to expand access to a broad range of treatment and recovery services, while keeping kids and communities safe,” she said.
The announcement follows two reviews of the sites that the government ordered in the wake of the killing of a Toronto woman, who was hit by a stray bullet from a shooting near one of the sites.
Karolina Huebner-Makurat had been walking through her southeast Toronto neighbourhood of Leslieville shortly after noon on July 7, 2023, when she was shot as a fight broke out between three alleged drug dealers.
The 10 sites that will have to stop offering supervised consumption no later than March 31, 2025 due to the new rules include five in Toronto, and one each in Ottawa, Kitchener, Thunder Bay, Hamilton and Guelph.
Nine of those sites are provincially funded and will be prioritized to receive funding under the new system so long as they give up supervised consumption services, Jones said.
The province will instead create 19 new “homelessness and addiction recovery treatment hubs” plus 375 highly supportive housing units at a cost of $378 million.
Those transitions would move quickly, she said.
The province said crime around these sites is “significantly higher” compared to surrounding neighbourhoods. It said reports of assaults are up 113 per cent and robberies up 97 per cent in neighbourhoods near the sites in Toronto compared to the rest of the city.
The new sites will not offer clean needles, Jones said.
A spokesperson for Toronto Mayor Olivia Chow said she is concerned the closures will lead to “increased overdose deaths, greater strain on first responders and emergency rooms, and more public drug consumption.”
A policy paper on opioid use from the Association of Municipalities of Ontario had the municipalities calling for a more collaborative approach and said the consumption sites save lives.
“A recent study of supervised consumption sites in Toronto found that a city-wide reduction in overdose mortality rate of 42 per cent after the implementation of supervised consumption sites,” the association wrote.
The South Riverdale Community Health Centre, near where Huebner-Makurat was shot, is among those that will no longer be able to offer supervised consumption and said it is reviewing the province’s decision.
“Our focus is on ensuring we have a plan in place for providing continued compassionate care to the clients we serve, support for our dedicated staff and dialogue with our neighbours,” said Gabriella Skubincan, a spokesperson for the health centre.
One site now set for closure in Ottawa was not consulted on the move.
“This will have devastating consequences for our community,” said Suzanne Obiorah, executive director of Somerset West Community Health Centre.
The Ford government introduced the consumption and treatment services model in 2018, saying it would focus on connecting people to treatment, rather than the previous supervised consumption model.
At that time, the province put in place a cap of 21 such sites in the province, but has only funded 17. There are other supervised consumption sites around the province that have received federal approval but no provincial funding, and two of them in northern Ontario recently closed due to a lack of funds.
Tuesday’s news from the government angered harm reduction workers, including one who works at one of the sites slated for closure.
“My heart sunk,” said Hannah Stahl, a registered nurse at one of the sites and co-lead of the Street Nurses Network. “My clients are going to be left in the lurch.”
She said she is able to provide health care, often daily, to her clients who otherwise are fearful of the health-care system. Stahl said she helps people get tested for various diseases and connects patients to counselling, helps them get on a list for housing or helps fill out forms to get identification.
“When people fall through the cracks of the system, this is what caught them,” she said.
But the pending closures are welcome to those living near South Riverdale Community Health Centre, where Huebner-Makurat was shot, said Derek Finkle.
He said he “completely agrees” with the province’s new approach to keep the sites away from schools.
Finkle, who lives across the street from the site and is a journalist who has written about the issue, said he has seen open drug use, fights, drug deals and tons of used needles. There are two schools nearby and six daycares, he said.
“One inevitable consequence of these sites is that it draws a number of drug dealers, some of whom carry guns and some of whom get into fights and then kill innocent passersby,” he said.
“It makes no sense that South Riverdale exists at an address that the province would prohibit from having a license to be a cannabis dispensary.”
Provincial regulations state cannabis shops must be no closer than 150 metres to a school.
Opioid deaths surged during the COVID-19 pandemic. In 2021, the mortality rate for opioid toxicity hit its peak of 19.3 deaths per 100,000 people, data from the Office of the Chief Coroner shows. That year, 2,858 people died from opioids, the vast majority of which contained fentanyl, a particularly potent opioid.
People dying from opioids are also increasingly found with benzodiazepines, cocaine, methamphetamine. And nearly five per cent of deaths in 2024 thus far contain xylazine, commonly used by veterinarians as a tranquilizer.
That mortality rate dropped to 17.5 deaths per 100,000 people, or 2,593 people, in 2023. Those numbers are significantly worse than pre-pandemic rates that saw 10.7 deaths, or 1,559 people, per 100,000, the coroner’s data shows.
The majority of opioid toxicity deaths, nearly 70 per cent, happen in private homes, the coroner’s data shows.
More than 600 people died of opioid toxicity in Ontario in the first quarter of this year, which is a slight drop from the same period in 2023.
For Jen la Fauci Gordon, a harm reduction worker who has been saved several times from overdoses at a supervised site, the government’s changes appear ideologically driven and will be deadly.
“It’s so callow to see those of us who quite often have the least political capital being used to score the easiest political points,” she said. “It’s disgusting.”
This report by The Canadian Press was first published Aug. 20, 2024.

by Grow Up Conference | Aug 21, 2024 | Grow Opportunity, Media Partners

by Grow Up Conference | Aug 20, 2024 | Cannabis News Wire, Media Partners
A ballot measure aiming to legalize recreational cannabis in North Dakota has been confirmed for the upcoming November election, as announced by the state’s chief election official on Monday. This development brings the issue back to voters, where previous attempts to pass similar measures have been unsuccessful.
Michael Howe, the state’s secretary of state, confirmed that almost 19,000 voter signatures were validated during the review process, surpassing the required number needed to secure a spot on the November ballot. The group responsible for the initiative, known as New Economic Frontier, had initially submitted more than 22,000 signatures in July.
The measure’s leader, Steve Bakken, former mayor of Bismarck and commissioner for Burleigh County, stated that law enforcement should concentrate more on battling fentanyl and opioids than cannabis. The initiative’s goal, he added, is to stop any actions taken outside of the state that might have unmanageable repercussions.
The proposed 20-page measure seeks to legalize recreational cannabis use for individuals 21 years of age and older. The measure also details various processing and production regulations and prohibited uses, such as in vehicles and public spaces.
The legislation sets limits on the amount of cannabis that can be purchased and possessed. Additionally, it would allow for edibles, cannabis solutions, transdermal patches, capsules and concentrates.
Currently in North Dakota, marijuana use by individuals under the age of 21 is classified as a misdemeanor. For those older than 21, recreational use is not illegal, but possession is, with penalties such as misdemeanors and infractions depending on the quantity of marijuana involved. Delivery of cannabis is considered a felony, with potential sentence enhancements if it occurs within 300 feet of a school.
Data from the state’s courts indicates that 4,451 individuals were charged with marijuana possession or use across the state last year.
While voters approved medical cannabis in 2016, they rejected recreational cannabis initiatives in both 2022 and 2018. In 2021, the state’s Republican-controlled House of Representatives passed measures to legalize recreational cannabis, but the Republican-controlled Senate ultimately defeated these efforts.
Representative Matt Ruby noted that the current priorities will be informing voters about the possible economic advantages, enhanced safety regulations and more convenient access to medical cannabis. On the other hand, the Brighter Future Alliance, a group opposing the measure, criticized supporters for not accepting defeat after multiple unsuccessful attempts.
Currently, recreational cannabis is legal in 24 states. Ohio was the most recent state to legalize marijuana in 2023 through a ballot measure. Voters in South Dakota and Florida will also have the opportunity to decide on similar measures in November.
In case the voters in these states give the nod to adult-use marijuana legalization at the polls, a chance will be created not only for marijuana companies but also ancillary ones such as Innovative Industrial Properties Inc. (NYSE: IIPR) to sprout in those markets and serve the entities that need their services.
About CNW420
CNW420 spotlights the latest developments in the rapidly evolving cannabis industry through the release of an article each business day at 4:20 p.m. Eastern – a tribute to the time synonymous with cannabis culture. The concise, informative content serves as a gateway for investors interested in the legalized cannabis sector and provides updates on how regulatory developments may impact financial markets. If marijuana and the burgeoning industry surrounding it are on your radar, CNW420 is for you! Check back daily to stay up-to-date on the latest milestones in the fast -changing world of cannabis.
To receive SMS alerts from CNW, text CANNABIS to 888-902-4192 (U.S. Mobile Phones Only)
For more information, please visit https://www.CannabisNewsWire.com
Please see full terms of use and disclaimers on the CannabisNewsWire website applicable to all content provided by CNW, wherever published or re-published: https://www.CannabisNewsWire.com/Disclaimer
CannabisNewsWire
Denver, CO
www.CannabisNewsWire.com
303.498.7722 Office
Editor@CannabisNewsWire.com
CannabisNewsWire is powered by IBN

by Grow Up Conference | Aug 20, 2024 | Media Partners, Psychedelic News Wire
Last week, the U.S. Food and Drug Administration (FDA) rejected a new drug application for MDMA to be administered as treatment for post-traumatic stress disorder. In its rejection, the federal agency asked the company that filed the application, Lykos Therapeutics, to conduct additional trials to further study the effectiveness and safety of its psychedelic formulation.
This would require additional funding as well as a number of years to complete, which doesn’t align with Lykos Therapeutics’ expectations.
The FDA’s ruling left many upset, particularly experts and patient advocates involved in the development and research of psychedelic treatments.
One neuroscientist at the University of California-San Francisco, Jennifer M. Mitchell, stated that the decision was a disappointment, adding that those involved in the application were confident they had met the criteria requested by the agency. Retired brigadier general Stephen Xenakis explained that these therapies were urgently required, not only by veterans but also other individuals experiencing similar problems.
Lykos Therapeutics is yet to make the letter of rejection available to the public. However, in a statement, the company stated that the issues voiced by the FDA were similar to those raised during an advisory committee meeting held a couple of months ago. The meeting was led by a panel of psychiatric experts who raised concerns about the safety and effectiveness of MDMA.
One of the issues raised was the inability to differentiate the psychedelic from the placebo effect as most participants in the trial could correctly guess whether they had received the placebo or the psychedelic.
Lykos revealed that it had responded to these issues, taking into account the suggestions raised and minimizing the effects of the issue.
Post-traumatic stress disorder affects roughly 13 million individuals in the United States, with current treatments only benefitting a fraction of these. The condition disproportionately affects veterans, with about 19% and 10% of female and male veterans respectively diagnosed with the disorder in 2021. Data from the Department of Veterans Affairs also shows that roughly 17 veterans die by suicide daily.
Prior to the FDA’s ruling, 730 veterans and 80 bipartisan legislators penned different letters to President Joseph Biden imploring him to approve the therapy.
Lykos plans to schedule another meeting with the agency where it will request that the FDA reconsider its ruling to conduct another trial. During the meeting, the company hopes to convince the agency to allow it to use existing data to file a new application instead. If the FDA declines this request, it is unclear what the company’s next step will be.
The entire psychedelics industry, including companies such as Compass Pathways PLC (NASDAQ: CMPS), will be anxiously waiting to see how this matter is concluded and any potential impact it may have on other psychedelics drug candidates under development.
About PsychedelicNewsWire
PsychedelicNewsWire (“PNW”) is a specialized communications platform with a focus on all aspects of psychedelics and the latest developments and advances in the psychedelics sector. It is one of 60+ brands within the Dynamic Brand Portfolio @ IBN that delivers: (1) access to a vast network of wire solutions via InvestorWire to efficiently and effectively reach a myriad of target markets, demographics and diverse industries; (2) article and editorial syndication to 5,000+ outlets; (3) enhanced press release enhancement to ensure maximum impact; (4) social media distribution via IBN to millions of social media followers; and (5) a full array of tailored corporate communications solutions. With broad reach and a seasoned team of contributing journalists and writers, PNW is uniquely positioned to best serve private and public companies that want to reach a wide audience of investors, influencers, consumers, journalists and the general public. By cutting through the overload of information in today’s market, PNW brings its clients unparalleled recognition and brand awareness. PNW is where breaking news, insightful content and actionable information converge.
To receive SMS alerts from PsychedelicNewsWire, text “Groovy” to 888-902-4192 (U.S. Mobile Phones Only)
For more information, please visit https://www.PsychedelicNewsWire.com
Please see full terms of use and disclaimers on the PsychedelicNewsWire website applicable to all content provided by PNW, wherever published or re-published: https://www.PsychedelicNewsWire.com/Disclaimer
PsychedelicNewsWire
San Francisco, CA
www.PsychedelicNewsWire.com
415.949.5050 Office
Editor@PsychedelicNewsWire.com
PsychedelicNewsWire is powered by IBN

by Grow Up Conference | Aug 20, 2024 | Grow Opportunity, Media Partners
By Tom Venetis
Cannabis Training Canada (CTC) says that its CTC 1: Retail Certification Program has been approved by the Alcohol and Gaming Commission of Ontario (AGCO) Board of Directors as an authorized cannabis retail employee training program.
Retailers and their employees will now have the option to use the CTC program to meet their mandatory training requirements. As an independent provider, Cannabis Training Canada’s says its program is free from affiliations with any cannabis producers or retailers, thereby ensuring and unbiased and comprehensive education for participants.
Read the full story here.

by Grow Up Conference | Aug 20, 2024 | Grow Opportunity, Media Partners
(Globe Newswire) Toronto – TerrAscend Corp., a leading North American cannabis company, today announced that its Board of Directors has authorized the Company to commence a normal course issuer bid (“NCIB”) to repurchase up to USD $10 million of the Company’s common shares, from time to time over a 12-month period.
Jason Wild, Executive Chairman of TerrAscend, stated, “Our first ever stock buyback program demonstrates our confidence in TerrAscend’s future and commitment to enhancing shareholder value. As famed investor Benjamin Graham famously stated, ‘in the short run, the market is a voting machine, but in the long run, it’s a weighing machine’. We are confident in the strength of our business, growth prospects, operational excellence, and strong cash flow. We believe our equity has compelling value and will be opportunistic with our share repurchases.”
There were 291 million Shares outstanding as of August 16, 2024. While the timeframe to purchase Shares starts on August 22, 2024, and ends no later than August 21, 2025, the Company is not obligated to purchase any Shares. If management determines it has a better use for its cash reserves, it is under no obligation to continue to purchase Shares and share purchases may be suspended or terminated at any time at TerrAscend’s discretion. The Company does not expect to incur debt to fund the share repurchase program.
The Company is authorized to repurchase up to 10,000,000 of the Company’s Shares, which represents 5% of the public float based on 291,513,055 total Shares outstanding as of August 16, 2024. There is a daily repurchase restriction of 65,361 Shares, which represents 25% of the Company’s average daily trading volume on the Toronto Stock Exchange of 261,445 Shares. Shares may be purchased on the Toronto Stock Exchange, the OTCQX Best Market, or alternative trading systems and will be subject to the limitations and rules imposed by U.S. and Canadian securities regulations. The actual number of Shares purchased, timing of purchases and share price will depend upon market conditions at the time and securities law requirements. All Shares acquired will be returned to treasury and cancelled.

by Grow Up Conference | Aug 20, 2024 | Grow Opportunity, Media Partners
Join cultivators Patrick Pagé of Great White North Growers and Mehgan Race Widdison of Mauve et Herbes as they share their commercial agriculture experiences in Quebec. Hear how their unique lifestyle, commitment to the craft and camaraderie with fellow growers propels the industry forward, creating a culture of continuous improvement and innovation in Canadian cannabis.

by Grow Up Conference | Aug 20, 2024 | Media Partners, Stratcann
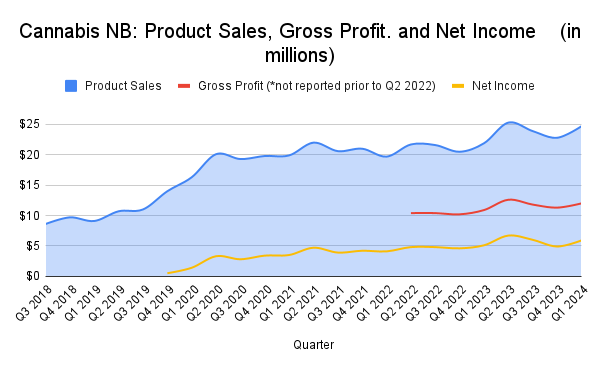
Cannabis sales bounced back in New Brunswick in the three months that ended June 30, 2024, following seasonal declines in the previous quarter from January through March.
Total product sales in the province for the quarter were $24.7 million, up 12.6% compared to the same quarter in the previous year. The provincial cannabis agency also brought in $5.9 million in net sales, up 15% from the same period in 2023.
Cannabis sales have continued to increase in the province annually, with consistent spikes in Q2, covering July, August and September, and declines in Q1, covering the first three months of the year.
Cannabis NB operates 27 stores in 18 communities in the province, along with nine private stores since the province began allowing them in 2023. There are also six cannabis farmgate locations in New Brunswick that allow cannabis producers to have a retail store on-site.
The agency’s 2024-2026 strategic plan says legal cannabis sales in the province have been approaching about 50% of the total cannabis market and includes plans to increase the number of retail outlets and products available, as well as exploring on-site consumption and customer loyalty programs.
Competition in the market has helped to bring prices down, continues the report, noting that while some of the big LPs are “struggling and trying to recover from over-investing” in incorrect market assumptions, “many new LPs and micro-producers are being licensed that have learned from the challenges at launch, and are approaching the industry with better information and a more sustainable plan.”
The organization also plans to establish a comprehensive loss prevention program to reduce risk. A Cannabis NB employee was recently charged with stealing more than $5,000 worth of products and cash. The case has been adjourned to September 6.

by Grow Up Conference | Aug 19, 2024 | Extraction Magazine, Media Partners
Like the instrumental evolution of classic column liquid chromatography, the HPLC takes advantage of the difference of affinity and interactions of each mixture component for the stationary and mobile phase leading to different retention times. This method found applications in various fields such as pharmaceutical, environmental sciences, and food science. [1]
High Performance Liquid Chromatography was developed in 1967 at Yale University, it is also known as high-pressure liquid chromatography because is characterized by using columns packed with very small particles, the stationary phase, and an eluting liquid phase, the mobile phase, pumped at high pressure through the column. [2]
The ability of the packed material to retain and separate compounds from a complex mixture is determined by functional groups chemically bonded to the surface of a support. The solute interacts with the functional groups via hydrogen bonds, dipole-dipole or electrostatic interactions. It has represented a significant advance in analytical science by revolutionizing the separation of complex extract mixtures. [2]
Importance of HPLC in Botanical Extraction
This technique offers high precision and accuracy that are essential for identifying and quantifying molecules. Because of its versatility, it can be used for the isolation of a wide range of botanical compounds including alkaloids, flavonoids, phenolics, and terpenes. Thanks to the possibility of standardizing operations, reproducibility is ensured, which is useful for quality control and suitable for rapid analysis (carried out in 30 minutes or less), in both research and industrial settings. [3]
Equipment
An HPLC consists of several assembled components. Generally, includes a sample filtration and eluent degassing system, containers for the eluent and for its discharge, an injection valve and possibly a sampler automatic, one or more pumps with their own controller, precolumn and column, one or more series detectors, and a data collection system. [2]
Materials
Samples and their preparation are primary for accurate analysis to identify and quantify the chemical constituents of leaves, stems, roots, seeds and flower extracts. The first step in specimen processing is extraction. The most commonly performed are extraction, supercritical fluid extraction and solid phase extraction. [4]
Once extracted, samples may need purification to remove interfering substances. This can be achieved by liquid-liquid extraction, filtration or centrifugation. The purified extracts are typically concentrated to improve detection sensitivity. High purity solvents and reagents are essential to minimize contamination and ensure reproducibility. [4]
To directly perform HPLC, the purified extract might have to be filtered and diluted with the mobile phase or an appropriate solvent. Filtration is carried out using special porous membranes, with holes that can reach 0.20 μm. Instead, if the sample is to be preserved will be stored in appropriate conditions, basically refrigerated and in the dark, to prevent degradation.
Eluent degassing is achieved by pumping the liquid under vacuum through membranes or allowing an inert gas such as helium to flow into the liquid, to remove dissolved air, which may collect in bubbles along the chromatograph circuit. [5]
The sample is introduced into the circuit using a syringe connected to a valve of injection. The purpose of this system is to allow the sample to enter the circuit while maintaining the latter, which is almost always under pressure, isolated from the outside.
Often the injection valve is part of an automatic sampler, which injects from time to time from the vials in which it is contained. Preparative HPLC pumps are often peristaltic, while most of the analytical HPLC pumps on the market are made up of a piston moved by a motor, and the flow direction is ensured by two check valves. The pump is equipped with a pressure gauge in the circuit. [5]
Selection of Columns and Solvents
The choice of columns is decisive and depends on the chemical nature of analytes. The columns can be housed in special compartments created in the pump container, and these are often thermostated to optimize the stationary phase performance.
The chromatographic circuit consists of small steel or PEEK (polyetheretherketone) tubes with an internal diameter of up to 0.25 mm. The precolumn is packed with the same stationary phase as the column, but has a lower length, and therefore cost. It serves to protect the column from possible damaging pollutants contained in the samples, which can bind to the stationary phase, decreasing its performance.
Normal-phase columns are suitable for polar compounds because the stationary phase, usually Silica gel, is more polar than the eluent used.
Reverse-phase columns are ideal for non-polar and moderately polar compounds because the stationary phase is apolar with respect to the eluent used. The most used are C18 columns, so called because the particles packed, made of silica gel or polystyrene-divinylbenzene, have hydrocarbon groups with 18 carbon atoms bound to the surface. The RP columns offer high resolution for a wide range of botanical compounds.
Ion exchange columns are used to separate ionic substances. The stationary phase is prepared by binding ionic functional groups to silica gel or to a porous support polymer (i.e. polystyrene); when the groups have a negative charge, the columns are called cation exchange, if they have a positive charge, they are called anion exchange. Aqueous buffers are used as eluent, possibly mixed with organic substances.
The strength of the eluent increases with the increase in the ionic strength of the buffer, species with a light charge will be eluted at lower concentrations, and those with a greater charge will be eluted as the concentration increases; furthermore, retention is influenced by the pH of the mobile phase and the content of organic solvent present.
Size exclusion columns are used to separate compounds based on sites of different sizes useful for larger biomolecules (proteins, DNA, etc).
The mobile phase should be chosen complementary to the column to increase the separation. It often consists of a mixture of water and an organic solvent such as acetonitrile or methanol supplemented with acids or buffers to improve peak shape and resolution. Gradient elution, in which the composition of the mobile phase changes over time, is efficient in complex mixtures. [5]
Optimization of Operational Parameters
Flow rate is one of the key parameters that affect the time that analytes spend in the column and their separation. A lower flow rate generally increases separation efficiency but extends time, while a higher flow rate reduces analysis time but may compromise resolution. The best parameter balance typically ranges from 0.5 to 1.5 mL/min. Adjustments are based on the specifications of columns provided by the manufacturer, and are related to the desired resolution. [6]
Another important parameter is the column temperature that has an influence in the interaction between the analytes and the stationary phase. Higher temperatures increase the speed but can lead to peak broadening.
Conversely, lower temperatures improve resolution but prolong analysis time. It is important to figure out the optimum, usually between 25 and 40°C, that provides the best compromise between the speed and resolution, keeping it constant to improve reproducibility. [6]
The injection volume must always be controlled to avoid overfilling the column, which can lead to peak distortion and loss of resolution. Typically, from 10 to 20 microliters, the volume depends on the column capacity and sample concentration. [6]
The detection wavelength is selected based on the absorption characteristics of the analytes.
- UV-Vis detectors are the most commonly used, and the wavelength is selected to maximize the response of the target compounds. This method is suitable for compounds with chromophores.
- Diode array detectors (DAD) permit to monitor multiple wavelengths simultaneously, providing more comprehensive information about the sample composition. DAD provides spectral information from which databases are created that allow the identification of unknown compounds while avoiding dereplication phenomena.
- HPLC can be performed in association with mass spectrometry (MS) that offers high sensitivity and specificity, allowing for the identification and quantification of trace compounds.
Data Collection and Analysis
First, the researcher should ensure that the HPLC system is configured correctly.
Start by degassing the mobile phase, then equilibrate the column by flowing the mobile phase through the column until a stable baseline is reached. By checking system parameters, verify flow, pressure and temperature settings according to the method protocol.
The run time must be set appropriately to guarantee that all compounds of interest elute from the column.
Data integration is performed using chromatographic software to integrate peaks and quantify compounds based on calibration curves. Peak identification is achieved by comparing retention times and spectral data with reference standards or literature values. [6]
After each analysis, don’t forget to clean the columns by washing with an appropriate solvent to remove residual compounds. Worn parts such as pump seals, injector seals and filters must be checked and replaced regularly. Periodic calibration with known standards and regular maintenance of the HPLC system helps preserve accuracy. [5]
Interpretation and Validation of Results
The interpretation of HPLC results involves analyzing chromatograms to identify and quantify the components of the sample. A chromatogram displays the detector response as a curve function of time, with each peak representing a different compound.
In fact, the retention time is a characteristic parameter that allows the compound that produced it to be unequivocally identified by comparing it with those of known standards. However, retention time alone may not be sufficient which is why it is combined with spectrometric analysis results for positive identification.
- Quantification is generally performed by measuring the area under the peak curve.
- Calibration lines, constructed using known concentrations of standards, are essential to balance the linear response of the detector over the range of concentrations encountered. [6]
Assessment of Parameters
Accuracy and precision are vital for reliable quantification. The former refers to how similar the measured values are compared to the actual value of the quantity of analytes in the sample. The second describes the regularity of responses when measurements are repeated under the same conditions. High precision means that repeated measurements produce similar results. [6]
HPLC method validation includes testing for linearity, which means that results (such as peak area or peak height) increase in direct proportion to the analyte concentration. This assures that as it takes more of a substance, the measurement reflects that increase. [6]
The smallest amount of an analyte that can be detected but not necessarily quantified is the limit of detection (LOD), it indicates the lowest concentration at which the analyte can be distinguished from the noise. Instead, the limit of quantification (LOQ) is the smallest amount of an analyte that can be quantitatively measured with acceptable precision and accuracy. It is higher than the LOD and ensures safe quantification. [6]
Robustness is the ability of the HPLC method to provide the same performance even with small variations in parameters like slight changes in temperature or flow rate and still produce great results. [6]
System Suitability Tests
These tests are conducted regularly to ensure the HPLC system is working correctly, and they include measurements of: peak symmetry which indicates good separation and proper functioning of the system; retention time repeatability by checking that the time it takes for an analyte to travel through the column remains consistent; theoretical plate count that is a measure of column efficiency, indicating how well the column separates. [6]
Troubleshooting
Now that it’s clear how complex this system is, let’s look at some troubleshooting.
Abnormal pressure in HPLC system can be an indicator of various problems:
- No pressure reading and no flow can be caused by air trapped in the pump or simply by the failure or shutdown of the instrument; obvious solutions are to degas the eluent and turn the instrument back on.
- Constant high pressure can result from excessive flow or blockage; solutions are to reduce the flow and possibly replace the porous septa.
- Constant low pressure is due to potential system leaks. [7]
System Leaks can be resolved with simple interventions: tighten or replace the fittings and replace the gaskets.
Problems with the chromatogram can significantly affect HPLC performance:
- Tailed peaks may indicate an exhausted column or clogged septa, in these cases it is possible to try turning and flushing the column or, at worst, replacing it.
- Split peaks are often caused by contamination of the pre-column or column inlet, here the only thing to do is replace the pre-column or column.
- Large, deformed peaks may be due to excess samples which can be eliminated by reducing the volume or concentration. [7]
Difficulties in inserting the injector may be due to the seals that need to be changed; if the sampler does not inject correctly there could be a lack of compressed air or electricity. [7]
Problems detectable with the senses (smell, sight and hearing) are indicative of mechanical wear: the smell is a symptom of leaks, high-pitched or grinding noises suggest poor lubrication or the need for replacement and maintenance. These interventions can extend the life of the system and minimize downtime. [7]
References:
- Foley, Joe P. “Liquid Chromatography: Theory and Methodology.” Analytical Chemistry (1996).
- Golay, M.J.E., & Kirkland, J.J. “Theory of chromatography and its application to high-performance liquid chromatography.” Journal of Chromatography (1965).
- B. Ramesh Kumar, “Application of HPLC and ESI-MS techniques in the analysis of phenolic acids and flavonoids from green leafy vegetables (GLVs)”, Journal of Pharmaceutical Analysis (2017).
- Mitra, Somenath, “Sample preparation techniques in analytical chemistry”. John Wiley & Sons (2004).
- [5] Reuhs, Bradley L. “High Performance Liquid Chromatography.” Food Analysis (2017).
- Indrayanto, Gunawan. “The importance of method validation in herbal drug research.” Journal of Pharmaceutical and Biomedical Analysis (2022).
- Dantu, Krishna & Sowmya, & Bhargavi, Sakala & Gopisetty, Surya & Sagar, Buthapalli & RAO, CH & Nama. Dr, Sreekanth. A REVIEW ON HPLC TROUBLESHOOTING. (2013)

by Grow Up Conference | Aug 19, 2024 | Cannabis News Wire, Media Partners
Marijuana is among the earliest crops cultivated by humanity, with ancient societies across the globe growing the plant for various purposes, including medicine, fiber and food as well as its hallucinogenic properties.
In the present day, approximately 12,000 years since marijuana’s first cultivation, the global marijuana industry has reached an estimated value of $65 billion. In regions where cannabis is legalized, consumers now have access to an extensive selection of more than 700 different strains, each with unique and often creative names, such as Unicorn Poop and Cat Piss.
Despite the wide range of options available, marijuana products are typically divided into two groups: sativa and indica, with each thought to have different psychotropic effects.
“There’s a common belief that smoking an indica strain is similar to using Xanax, leading to drowsiness, while consuming sativa is akin to a stimulating experience, almost like taking a hit of cocaine, resulting in increased energy and hyperactivity,” explains Binske cofounder and CEO, Alex Pasternack.
However, a growing body of scientific evidence indicates that these names may not imply as much as previously believed. There may not be many genetic or chemical distinctions between the two, suggesting that users may not get the desired results.
French scientist Jean-Baptiste Lamarck is credited with coining the words indica and sativa in the late 1700s when he proposed classifying marijuana plants according to their physical traits. He noted that sativas were longer with fibrous stems and narrow, thin leaves, whereas indicas were shorter with woody stems and broad, stubby leaves.
However, neuroscientist Nick Jikomes, who was once Leafly’s director of science and innovation, states that a plant’s outward look is not always a good sign of its effects on the mind and body. He says that a drug’s chemical makeup determines how it affects you.
And unlike wineries, cheesemakers or apple producers, cannabis farmers are not constrained by rigid naming conventions. According to Jikomes, anyone may take any random cannabis seed and call it whatever they wish. They can also classify it as indica, sativa, hybrid or anything else they want.
This helps to explain why Jikomes discovered that product labels frequently misrepresented the chemical makeup of the marijuana flowers it tested in a 2022 study that examined the chemical composition of more than 90,000 commercial marijuana flower samples from six states in the country. The study found that sativas did not consistently contain higher levels of THC, the primary compound responsible for the marijuana high, compared to indica strains.
These findings are consistent with a study conducted in 2021, which examined around 300 marijuana strains. The lead author of the study, Sean Myles, stated that there isn’t any credible scientific data indicating a difference between samples classified as indica or sativa.
The widespread cross-breeding that has taken place throughout time is partially to blame for the confusion. Myles noted that although the original indica plant, which originated in the Middle East, may have been chemically and genetically different from its sativa counterpart from central Asia, the differences are now too hazy to be distinguished with clarity.
Researchers recommend categorizing marijuana according to its chemical properties rather than its physical characteristics for increased accuracy. Cannabis products would be labeled by their primary chemical constituents and their concentrations, much like the Nutrition Facts label on food. Ingredients would be the biologically active compounds produced by the plants, such as terpenes and cannabinoids.
While a labeling system based on the chemical composition of a strain may be perfect for established marijuana markets, Pasternack acknowledges that most consumers today are not seeking scientifically accurate or more detailed labels. They generally look for the best value for their money, he says.
Industry actors such as Green Thumb Industries Inc. (CSE: GTII) (OTCQX: GTBIF) have plenty of work to do to make customers more aware of how different cannabinoids combine to provide the desired effects, regardless of whether the strain is indica or sativa.
About CNW420
CNW420 spotlights the latest developments in the rapidly evolving cannabis industry through the release of an article each business day at 4:20 p.m. Eastern – a tribute to the time synonymous with cannabis culture. The concise, informative content serves as a gateway for investors interested in the legalized cannabis sector and provides updates on how regulatory developments may impact financial markets. If marijuana and the burgeoning industry surrounding it are on your radar, CNW420 is for you! Check back daily to stay up-to-date on the latest milestones in the fast -changing world of cannabis.
To receive SMS alerts from CNW, text CANNABIS to 888-902-4192 (U.S. Mobile Phones Only)
For more information, please visit https://www.CannabisNewsWire.com
Please see full terms of use and disclaimers on the CannabisNewsWire website applicable to all content provided by CNW, wherever published or re-published: https://www.CannabisNewsWire.com/Disclaimer
CannabisNewsWire
Denver, CO
www.CannabisNewsWire.com
303.498.7722 Office
Editor@CannabisNewsWire.com
CannabisNewsWire is powered by IBN









Recent Comments